|
6/10/2023 0 Comments Nh3 acid or baseSo although NH3 is technically a weak base, it can act as a base and acid depending upon the situations and the molecules it is reacting with. It combines with acids to form ammonium salts thus with hydrochloric acid it forms ammonium chloride (sal ammoniac). Here as Ammonia has a lone pair of electrons and accepts a proton, it is considered as a Bronsted-Lowry Base. According to Bronsted-Lowry theory, any compound that accepts a proton is considered as Bronsted-Lowry base. Whereas under certain conditions, NH3 can act as a weak acid and donate one H+ that forms its conjugate base NH2+. Here although Ammonia is a weak base, it is amphoteric as it can act as an acid as well as a base depending upon the conditions in which the experiments are conducted.Īmmonia can act as a weak base under a suitable condition and accepts H+, which results in forming NH4+, a conjugate acid. Generally, the compounds having a pH between 7 to 14 is base. Is NH3 acidic or basic?Īmmonia has a pH 11, which makes it a weak base. 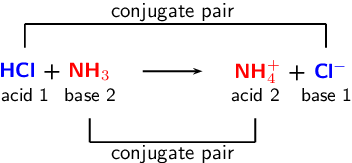
Keep reading this blog post to find out if NH3 is an acid or base. It can lose H+ ion and form Amides (NH2-). Ammonia is different from other molecules, and hence one-word answer for this question wouldn’t suffice. Although NH3 is a weak base, it also acts as a weak acid under certain conditions and reacts with bases. 
Generally, a molecule is classified as a base or acid by looking at its pH. It is vital to know whether this molecule is a base or acid to understand its chemical properties further. Ammonia is also used in manufacturing dyes, pesticides, and other such chemicals. It has a trigonal bipyramidal molecular geometry and one of the known components in agriculture as it is widely used as a fertilizer. The distribution of electrons in Ammonia is asymmetric, which is why it is also a polar molecule. It is made up of one nitrogen atom and three hydrogen atoms. It has a pungent smell and a colorless, non-flammable gas. 
Ammonia or NH3 is one of the interesting compounds to study about.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
